Sci-fi movies have often fascinated us with the superhuman potential that the switching 'on and off' of genes can give rise to. However, in the real world, the expectation from this process is more modest, say tackling diseases. Opening new doors in this paradigm, scientists have combined CRISPR technology and artificial intelligence (AI) to develop a technique where individual genes can be awakened through the disabling of chemical "off switches" that silence or suppress them.
In a new multi-institutional study, scientists have used AI to create a protein, and then apply CRISPR technology to it in order to inhibit a protein complex called Polycomb Repressive Complex 2 (PRC2) to awaken and upregulate several genes. According to the researchers, the new approach will aid in understanding the role that individual genes play in normal functions such as cell growth and development, processes such as aging, and diseases such as cancer, better.
"The beauty of this approach is we can safely upregulate specific genes to affect cell activity without permanently changing the genome and cause unintended mistakes," said Dr. Shiri Levy, lead author of the study, in a statement. The research was published in the journal Cell Reports.
A Novel Approach

What makes this technique unique is that the activity of the genes is controlled without any alteration to the DNA sequence of the genome. This is done by targeting chemical modifications that aid in the packaging of the genes within the human chromosomes and moderate their activity. As these changes occur on top of the genes and not inside them, they are known as 'epigenetic' (Greek: epi means ''above' or 'over') changes. Therefore, the chemical modifications that control gene activity are known as epigenetic markers.
Epigenetic modifications are of particular interest to scientists because their effect extends beyond that on gene activity within the realm of normal cell function. The accumulation of epigenetic markers over time promotes aging. In addition to this, they can also have a profound effect on the health of one's offspring and future generations as they can be passed on from parent to child.
Blocking Key Protein Complex
In the study, the authors concentrated on Polycomb Repressive Complex 2 (PRC2), a protein complex. While PRC2 is active throughout human development, it plays a crucial role, particularly during the embryonic stage where embryonic cells metamorphose into several cell types that result in the formation of tissues and organs within the growing embryo.

PRC2 suppresses genes by adding a methyl group to a protein responsible for the packaging of genes called histones. Methyl groups are structural units of organic compounds. If PRC2 is blocked, then the genes it has suppressed can be awakened again. Chemicals can be utilized to block PRC2. However, they are not reliable. Therefore, the team strived to find a method that could block PRC2 in a manner that affected only one gene at a time.
In order to accomplish this, the researchers used AI to create a protein called EED binder (EB) that could attach itself to PRC2 and hinder the protein that PRC2 utilizes to alter histones. Following this, the team fused the fabricated protein with a deactivated form of a protein called Cas9.
Modifying A Crucial Protein
In CRISPR, a gene editing technique, Cas9 binds with RNA and uses it as an 'address tag'. This process enables scientists to guide Cas9 to specific locations in the genome by synthesizing particular 'address-tag' RNA. As a result, genes can be cut and spliced at specific locations. For this study, the slicing action of Cas9 was disabled. Therefore, the genomic DNA sequence remained unaltered; resulting in 'dead' Cas9 or dCas9.
Nevertheless, the function of Cas9 as a vehicle to carry cargo to a particular location in the genome remained unchanged. The AI-designed blocking protein EB served as the cargo for the dCas9-RNA construct. Simplifying the process, Dr. Levy explained, "dCas9 is like UBER. It will take you anywhere on the genome you want to go. The guide RNA is like a passenger, telling the UBER where to go."
Turning On Individual Genes
Utilising the new technique, the scientists demonstrated that it was possible to block PRC2, and four different genes were selectively turned on. In addition to this, they also showed that induced pluripotent stem cells could be converted to placental progenitor cells by merely turning two genes on.
"This technique allows us to avoid bombarding cells with various growth factors and gene activators and repressors to get them to differentiate. Instead, we can target specific sites on the gene transcription promoters' region, lift those marks and let the cell do the rest in an organic, holistic manner," illustrated Dr. Levy.
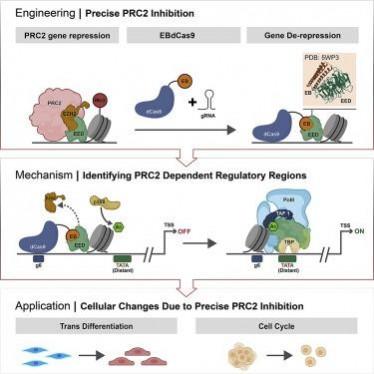
Also, the team was able to demonstrate that the technique can be leveraged to locate specific PRC2-controlled regulatory regions from where activation of individual genes takes place. Interestingly, the locations of several of these regions remain unknown. Through the current research, the authors recognized a promoter region—known as a TATA box—for a gene called TBX18, which is associated with several diseases.
Question Accepted Norms
This step led to the challenging of the general understanding that these promotor regions are near the gene, mostly within 30 DNA base pairs (building blocks of the DNA double helix). For the TBX18 gene, the promoter region was found to be over 500 base pairs away.
Dr. Hannele Ruohola-Baker, lead author of the study, noted, "This was a very important finding. TATA boxes are scattered throughout the genome, and current thinking in biology is that the important TATA boxes are very close to the gene transcription site and the others don't seem to matter. The power of this tool is that it can find the critical PRC2 dependent elements, in this case, TATA boxes that matter."
Importantly, the findings of the study shed more light on epigenetic modifications. "With these two advances, AI-designed proteins and CRISPR technology, we can now find the precise epigenetic marks that are important for gene expression, learn the rules and utilize them to control cell function, drive cell differentiation and develop 21st-century therapies," concluded Dr. Ruohola-Baker.

















